Electromagnetic Tracking for Image-Guided Urology
As urologic procedures evolve across prostate interventions, and stone management, OEMs are integrating Electromagnetic (EM) tracking to support real-time localization of instruments within and around soft tissue anatomy. Aurora® EM tracking from NDI provides continuous position and orientation data for integration into OEM-developed systems supporting image-guided urologic workflows.
OEMs Navigate with Anatomical Imaging and Continuous Instrument Tracking with Aurora®
Aurora provides the real-time localization layer that connects pre-procedure MRI or CT imaging, ultrasound visualization, and continuous instrument tracking inside OEM-integrated urology workflows.
Pre-Planning
OEM Tools for Needle Trajectory. Aurora provides positional data. OEM platforms using tracking data alongside MRI or ultrasound imaging may enable trajectory visualization capabilities.
Instrument-to-Target Localization
Real-Time Position Data for Biopsy. Aurora provides continuous positional data for biopsy needles and other instruments relative to MRI or ultrasound-derived anatomical structures.
Image Fusion
EM Data for Multi-Modality Integration. Aurora supplies real-time spatial data that OEM platforms can use to register and maintain alignment between pre-procedural imaging datasets and live ultrasound imaging.
Electromagnetic Tracking for Urology
Aurora integrates into OEM urology systems to provide real-time instrument localization across biopsy, focal therapy, and percutaneous workflows.

Transrectal & Transperenial Ultrasound Probe
In OEM imaging systems, an EM tracked probe can be used to track depth, image fusion and relative needle placement.
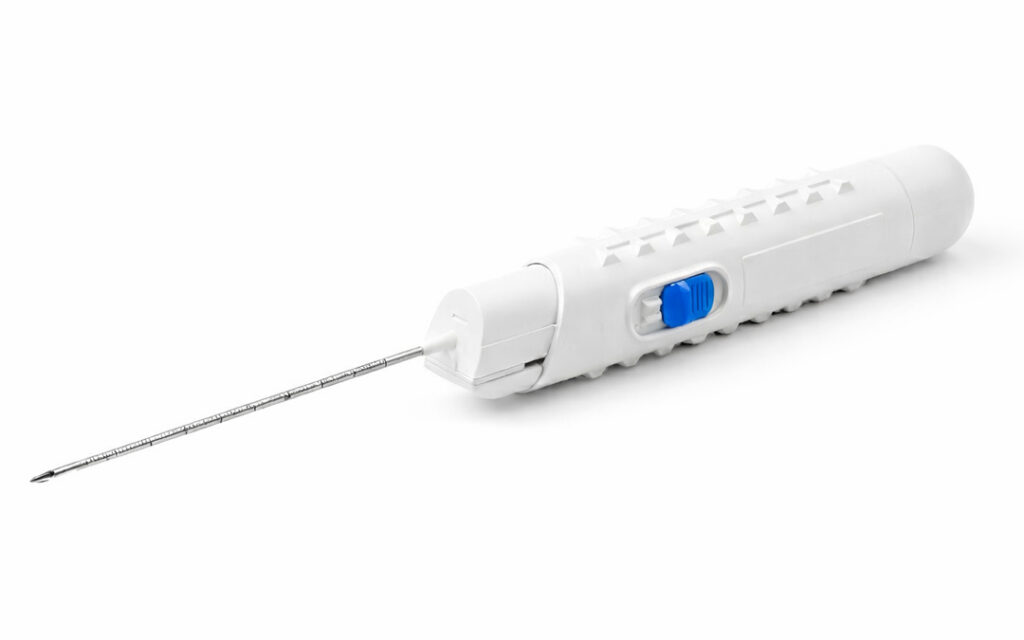
Biopsy Needle
When paired with OEM imaging systems, EM tracked biopsy needles can be used to provide position and trajectory data.
Recommended Products

Aurora Electronics Units
Designed for low-cost 5DOF tracking of disposable tools. PCB version designed for integration directly into OEM imaging carts.

Aurora Planar 20-20 V2 or X2
Mounts to bedside, OEM ultrasound cart, or separate stand that rolls in and out of the procedure when needed. Extended volume for larger tracking spaces.
Aurora Sensors

Transrectal Ultrasound Probe sensor
Popular Choice: 6DOF PCB Sensor
Ultrasound probes can accommodate larger sensors like PCB sensors, which offer the highest accuracy.
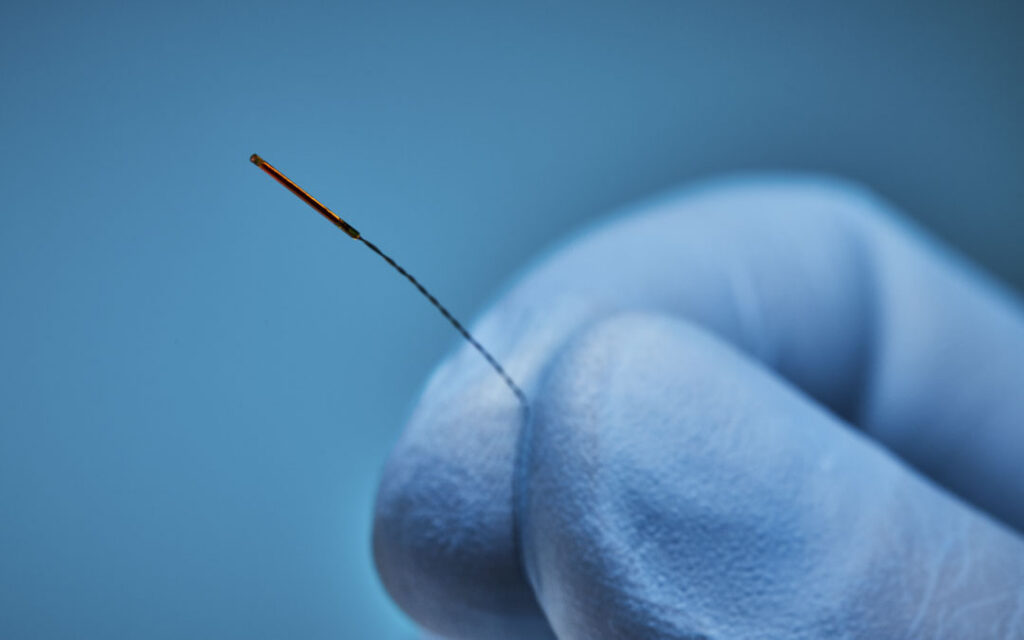
Biopsy Needle Sensor
Popular Choice: 5DOF Solid Sensor
Small, low cost sensors are critical for disposable needle workflows. 5DOF provides the smallest sensors to date.
Developing an Image-Guided Urology System?
Connect with our Product Integration team to identify the right Aurora tracking configuration for your prostate biopsy, focal therapy, or percutaneous navigation platform.
Frequently Asked Questions: Electromagnetic Tracking for Urology
Why does tracking matter in urology?
Urologic interventions increasingly rely on ultrasound, MRI, and fluoroscopy to guide procedures within the prostate, kidney, and surrounding anatomy. While these imaging methods provide critical anatomical context, they do not continuously represent instrument position as instruments advance through tissue. Electromagnetic tracking addresses this gap by providing continuous, real-time localization of instruments relative to patient anatomy and pre-procedural imaging. For OEMs developing urology platforms, EM tracking provides data that supports: MRI-ultrasound fusion for targeted prostate biopsy, digital mapping of biopsy core locations, multi-probe spatial coordination for focal therapy, and real-time needle localization for transperineal access. As clinical demand grows for targeted biopsy, transperineal access, and focal therapy alternatives, continuous instrument tracking is becoming a foundational capability in urology system design.
What types of urologic procedures does NDI electromagnetic tracking support?
NDI Aurora supports OEM development of image-guided systems for MRI-ultrasound fusion prostate biopsy, transperineal prostate biopsy, and prostate focal therapy procedures including HIFU, cryotherapy, irreversible electroporation (IRE), and laser ablation. Aurora is also used in OEM systems designed for percutaneous kidney procedures where instrument tracking supports access planning and probe placement.
How does Aurora support MRI-ultrasound fusion biopsy?
Aurora provides continuous, real-time position and orientation data for the ultrasound probe and biopsy instruments. This enables spatial registration between pre-procedural MRI and live ultrasound, supporting lesion targeting workflows, biopsy core mapping, and procedure documentation within OEM-integrated systems.
Why is electromagnetic tracking used in urology instead of optical tracking?
Urologic procedures typically involve instruments operating inside or around soft tissue anatomy where direct line of sight is not available. Electromagnetic tracking does not require line of sight between the sensor and the tracking system, making it suitable for tracking biopsy needles, ablation probes, and ultrasound transducers during procedures where the instrument is inside the body.
Which Aurora field generators are recommended for urology procedures?
NDI offers multiple options. The Planar 20-20 mounts bedside for stable tracking during biopsy and focal therapy. The Planar 10-11 is compact and handheld for use near the target region. The Window Field Generator sits beneath or behind the patient, keeping the workspace clear. The choice depends on the OEM’s system design and workflow.
What is the smallest sensor NDI offers for urologic instruments?
Aurora supports sensors with diameters as small as 0.3 mm, enabling integration into biopsy needles, ablation probes, and other instruments where minimal sensor size is critical. Both 5DOF and 6DOF configurations are available.
Can Aurora track multiple instruments simultaneously during focal therapy?
Yes. Aurora supports simultaneous tracking of multiple sensors, which is relevant for focal therapy procedures that involve coordinating the placement of multiple ablation probes. Real-time position data for each probe is one input that OEM-integrated systems may use within their spatial alignment and treatment planning workflows, as defined by the OEM system’s intended use.